Neurological disease and cognitive dynamics (II): Neural oscillations and cognitive dynamics
-
摘要: 大脑神经系统具有从慢到快多种不同的振荡节律, 这些节律振荡被认为参与了大脑多种功能的实现, 其中高频的伽马同步振荡被认为与大脑的认知功能最为相关. 本文阐述了生物学实验方面关于伽马振荡及其功能的研究进展, 并针对实验中伽马振荡的频率敏感依赖于外部刺激特征的现象, 综述了基于神经网络模型进行变频伽马振荡及其认知功能的动力学建模研究工作, 解释了视觉刺激调控的变频率伽马振荡动力学产生机理, 提出了基于同步抑制增强全局放电率对比度的神经认知机制. 研究成果有助于理解神经系统同步振荡的产生机理及其认知作用, 为大脑认知原理以及类脑智能的研究奠定基础.Abstract: The brain nervous system has various oscillatory rhythms, from slow to fast. These rhythmic oscillations are believed to be involved in the realization of various brain functions. The high-frequency Gamma synchronous oscillations are considered to be most related to the cognitive functions of the brain. In this review paper, the research progress of Gamma oscillations and their functions in biological experiments is expounded. Then, concerning the biological observation that the frequency of Gamma oscillations sensitively depends on the characteristics of external stimuli, the dynamical modeling work on the variable-frequency Gamma oscillations and the cognitive functions based on neural network models is also expounded. In this paper, the generation mechanisms of variable-frequency Gamma oscillation dynamics regulated by visual stimuli are explained, and a neurocognitive mechanism of global enhancement of firing rate contrast based on synchronous inhibition is proposed. The research results are helpful to understand the generation mechanisms of synchronous oscillations of nervous system and the cognitive functions and lay a foundation for the study of brain working mechanisms of cognitive activities and brain-like intelligence.
-
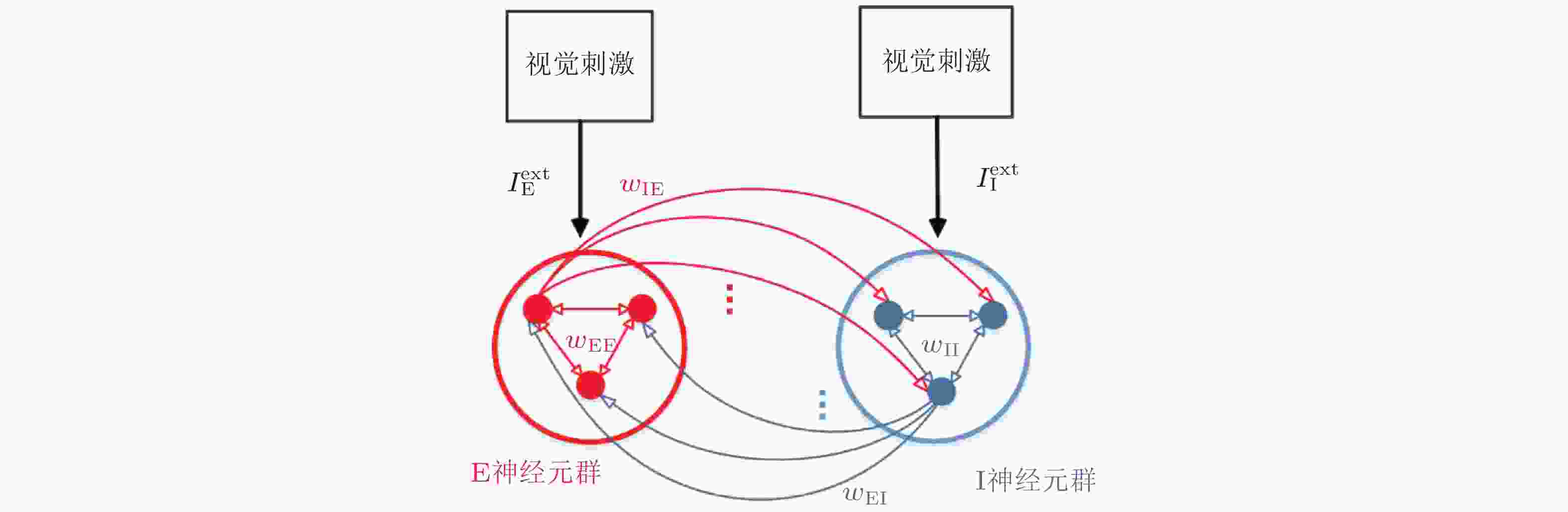
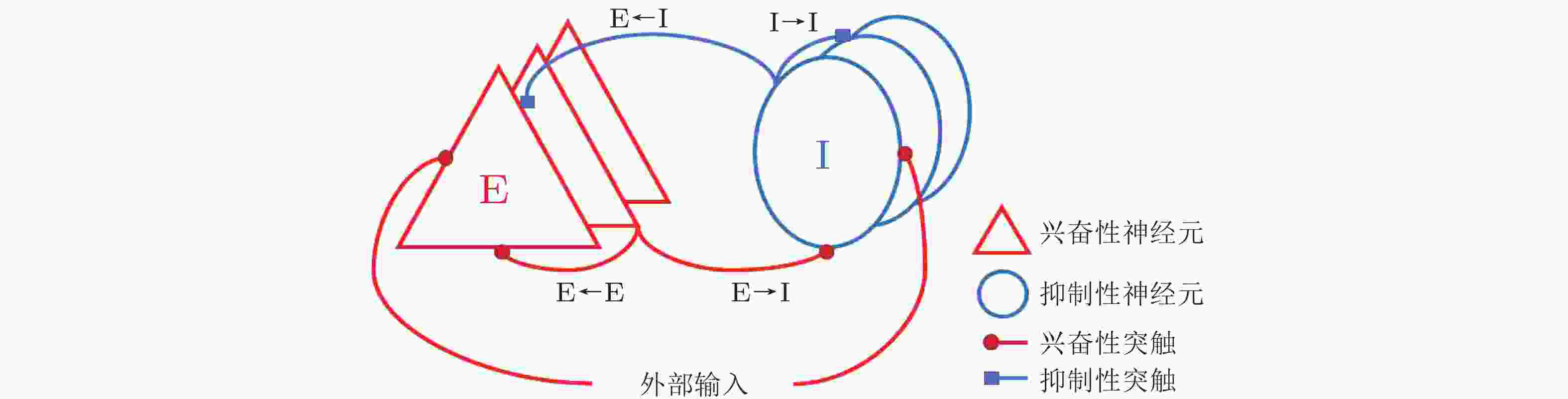
图 2 简单刺激调控的E/I神经网络. 红色和蓝色的圆分别表示E、I神经元集群 (400个兴奋性神经元和100个抑制性神经元) , 红色定向线表示兴奋性连接, 蓝色定向线表示抑制性连接.
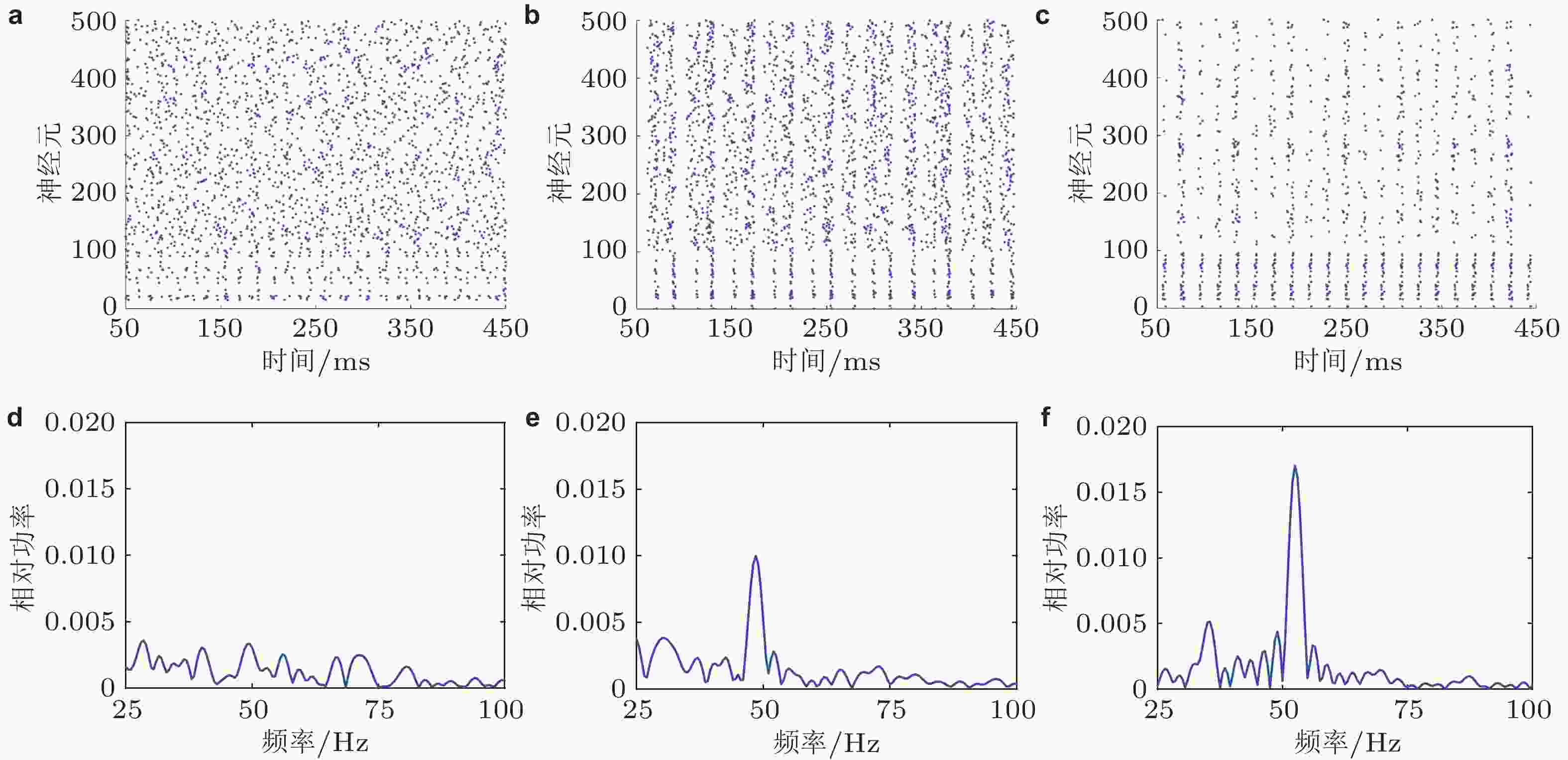
$ {w}_{\mathrm{E}\mathrm{E}} $ 、$ {w}_{\mathrm{I}\mathrm{I}} $ 、$ {w}_{\mathrm{E}\mathrm{I}} $ 和$ {w}_{\mathrm{I}\mathrm{E}} $ 分别是E神经元连接到E神经元、I神经元连接到I神经元、E神经元连接到I神经元、I神经元连接到E神经元的突触权重.$ {I}_{\mathrm{E}}^{\mathrm{e}\mathrm{x}\mathrm{t}} $ 和$ {I}_{\mathrm{I}}^{\mathrm{e}\mathrm{x}\mathrm{t}} $ 分别表示网络中E神经元和I神经元接收到的外部刺激输入 (Gu et al. 2021a)图 3 平衡和具有外部输入差异的E/I神经网络的仿真结果.左列、中列、右列图分别为
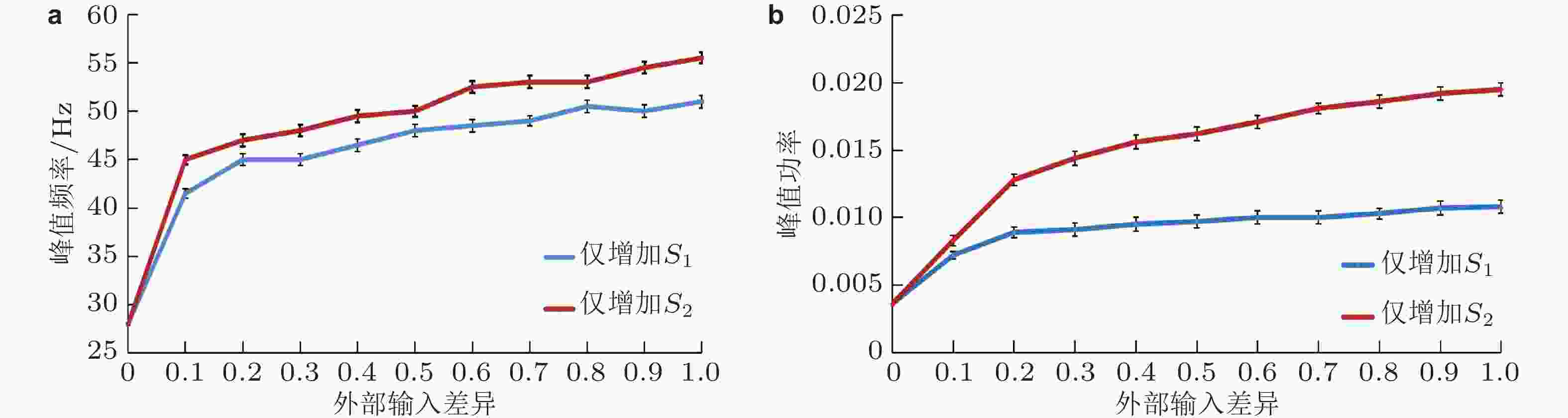
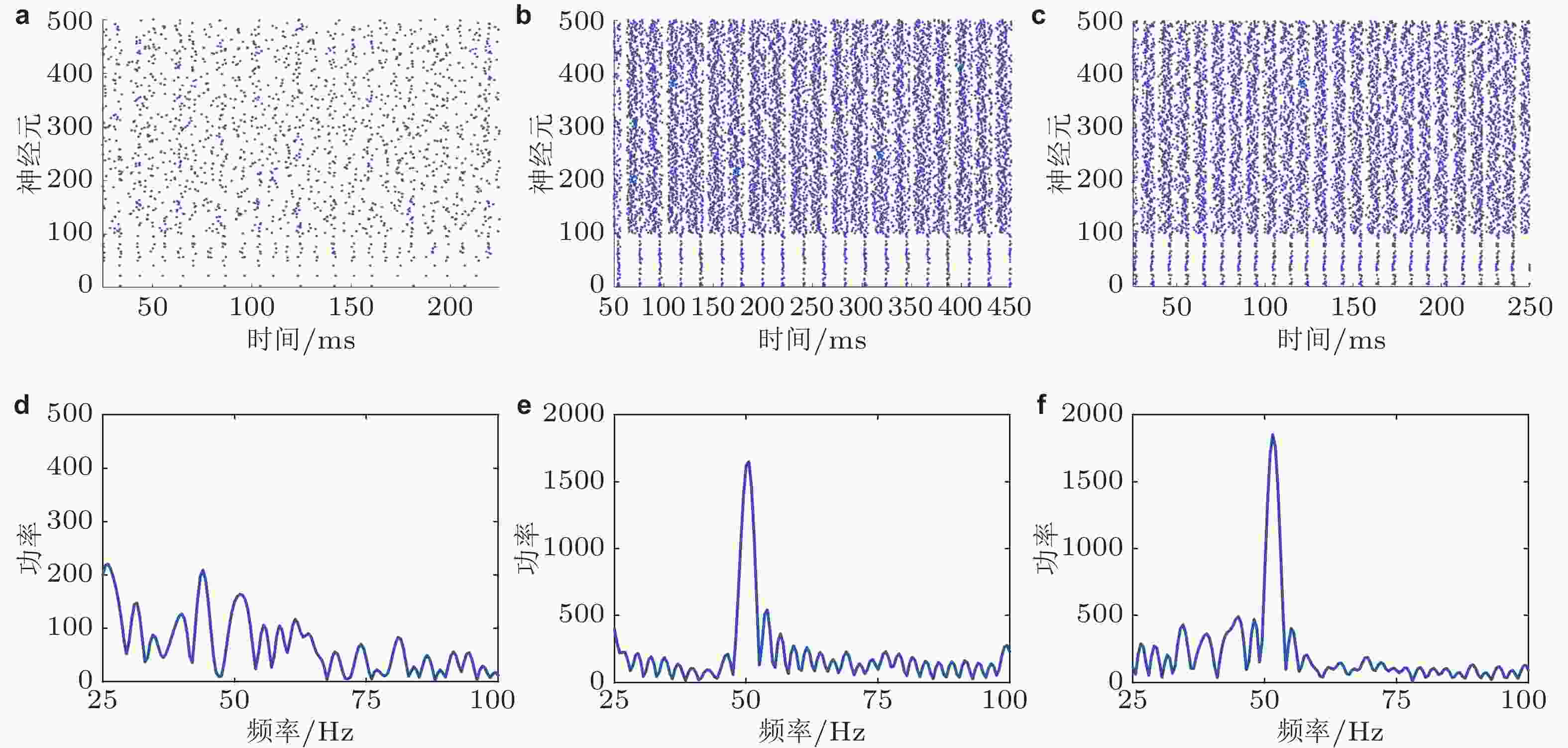
$ \mathrm{\Delta }S=0 $ (S1 = S2 = 2.5 μA) 、$ \mathrm{\Delta }S < 0 $ (S1 = 2.5 μA, S2 = 3.1 μA) 和$ \mathrm{\Delta }S > 0 $ (S1 = 3.1 μA, S2 = 2.5 μA) 的情况. (a) (b) (c)神经元的放电时刻斑图, 编号为0 ~ 100的神经元是抑制性神经元, 编号为101 ~ 500的神经元是兴奋性神经元; (d) (e) (f)神经元平均群体活动的功率谱图 (这里采用的是相对功率, 即功率谱中每个频率分量的功率与所有频率分量的功率之和的比值) (Gu et al. 2021a)图 4 两种刺激调节方式下Gamma振荡和E、I神经元集群的外部输入差异之间的关系 (中心刺激对比度增加由增大S1、保持S2不变来实现, 周围刺激对比度增加由增大S2、保持S1不变来实现). (a) Gamma振荡的频率随外部输入差异的增大而增大; (b) Gamma振荡的峰值功率随外部输入差异的增大而增强 (Gu et al. 2021a)
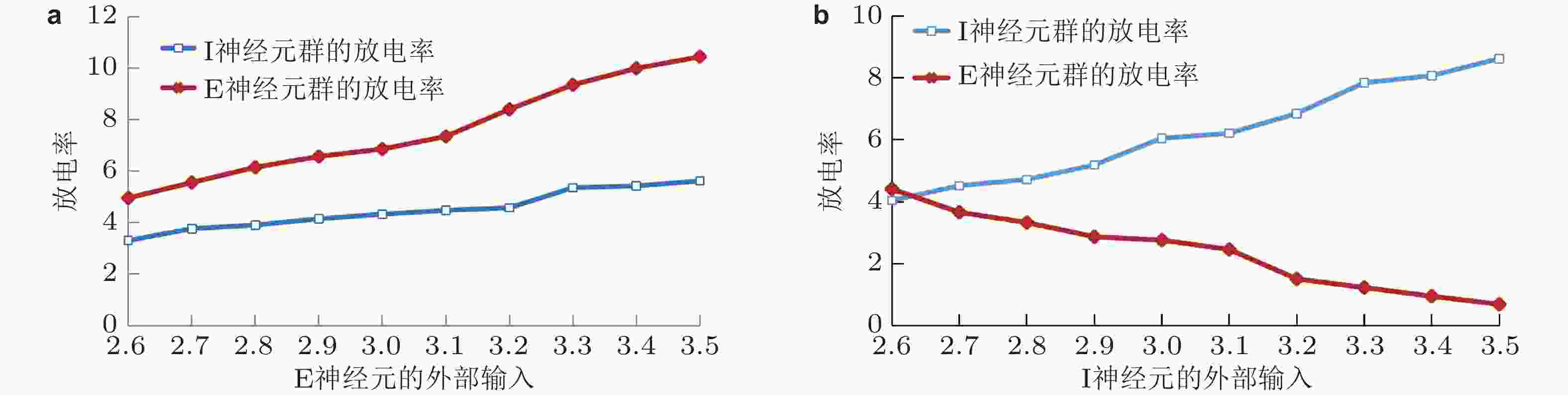
图 5 E/I 神经网络中 E 神经元集群和 I 神经元集群的平均放电率. (a) E 神经元具有不同输入时 E, I 神经元集群的放电率变化; (b) I 神经元具有不同输入时 E, I 神经元集群的放电率变化 (Gu et al. 2021a)
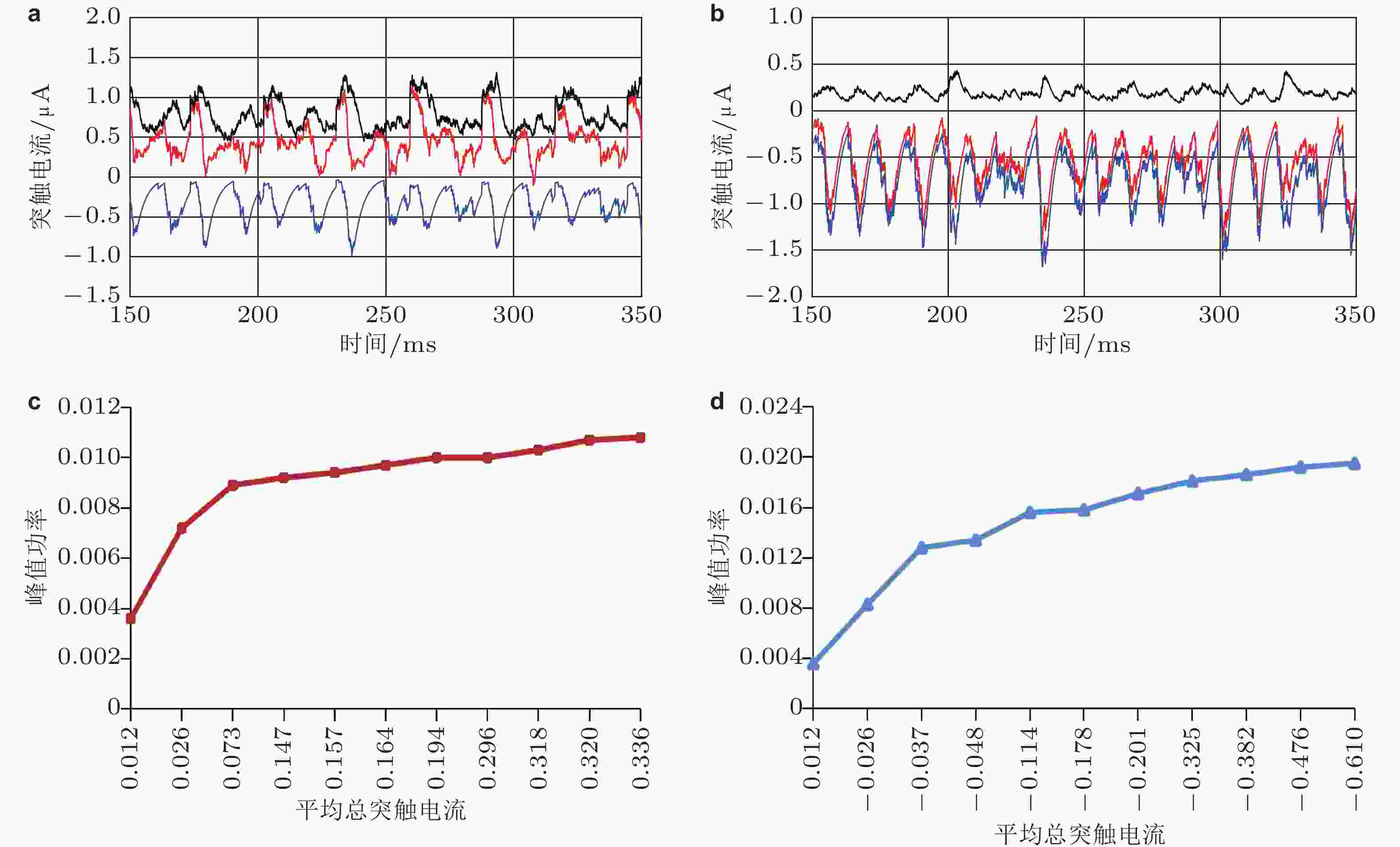
图 6 E/I 神经网络中任意一个神经元的突触电流: (a)
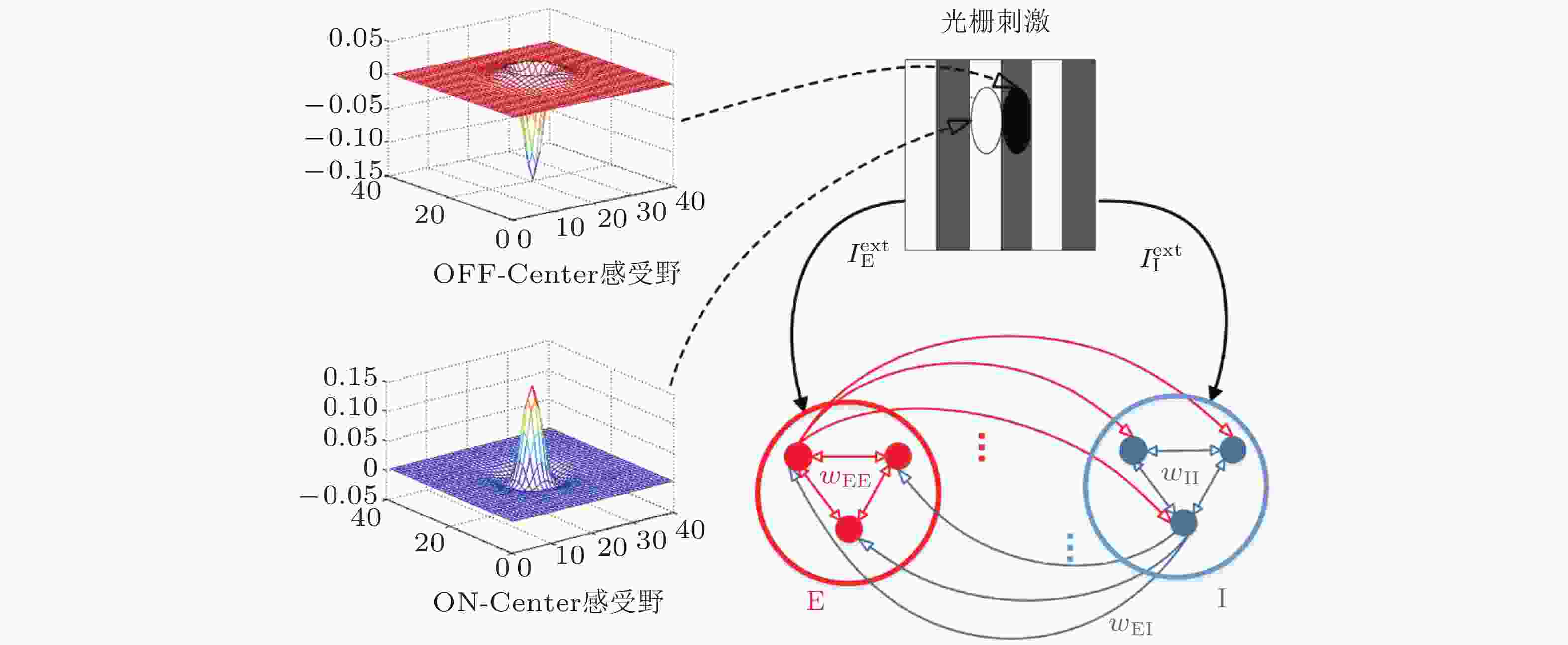
$ \mathrm{\Delta }S > 0 $ 的情形 (E 神经元和I 神经元的输入分别为 3.5 μA和 2.5 μA) ; (b)$ \mathrm{\Delta }S < 0 $ 的情形 (E 神经元和I 神经元的输入分别为2.5 μA和3.5 μA) . 图中黑色线表示该神经元接收到的兴奋性突触电流, 蓝色线表示该神经元接收到的抑制性突触电流, 红色线表示总突触电流, 为前二者之和. 网络平均总突触电流和伽马振荡的峰值功率之间的关系: (c)$ \mathrm{\Delta }S > 0 $ 的情形 (E 神经元和I 神经元的输入分别为 3.5 μA和 2.5 μA) ; (d)$ \mathrm{\Delta }S < 0 $ 的情形 (E 神经元和I 神经元的输入分别为2.5 μA和3.5 μA) (Gu et al. 2021a)图 7 视觉刺激调控的E/I神经网络的结构, 左侧示意图分别表示E神经元 (下方) 和I神经元 (上方) 的感受野的空间结构. 叠加在光栅刺激 (右上方) 上的感受野显示为两个椭圆区域, 黑色的表示OFF区域, 白色的表示ON区域 (Gu et al. 2019)
图 8 光栅无灰度对比度的平衡E/I神经网络 (左列图) 、第一种调节方式下光栅具有40%灰度对比度的E/I神经网络 (中列图) 、第二种调节方式下光栅具有60%灰度对比度的E/I神经网络 (右列图) 的仿真结果. (a) (b) (c) 神经元的放电时刻斑图, 标号0 ~ 100的神经元是抑制性神经元, 标号101 ~ 500的神经元是兴奋性神经元; (d) (e) (f) 神经元平均群体活动的功率谱图 (Gu et al. 2019)
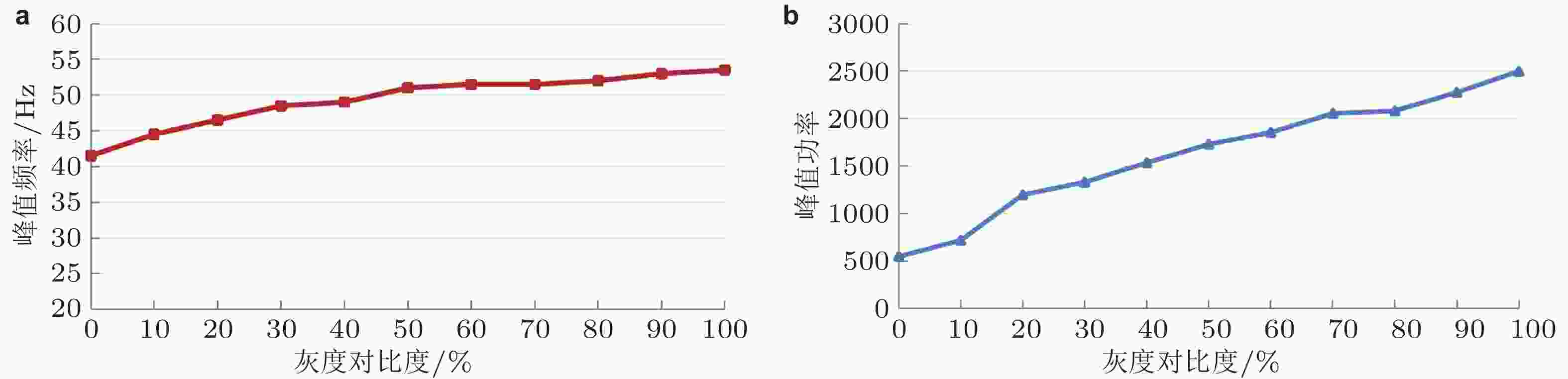
图 9 光栅不同灰度对比度引发Gamma振荡的 (a) 峰值频率和 (b) 峰值功率 (灰度对比度调节方式: 同时增加亮条纹的灰度值
$G_1$ 和减小暗条纹的灰度值$G_2$ ) (Gu et al. 2019)图 10 具有立柱结构的大规模复杂E/I神经网络. (a) 单个立柱中层内和层间神经元之间的连接; (b) V1 区的简化模型 (Kang et al. 2010) , 其中 E 表示兴奋性神经元集群, I 表示抑制性神经元集群, 箭头表示神经信号投射方向, 图中只画出 2 个功能柱; (c) 立柱和立柱之间神经元的连接结构 (Gu et al. 2021b)
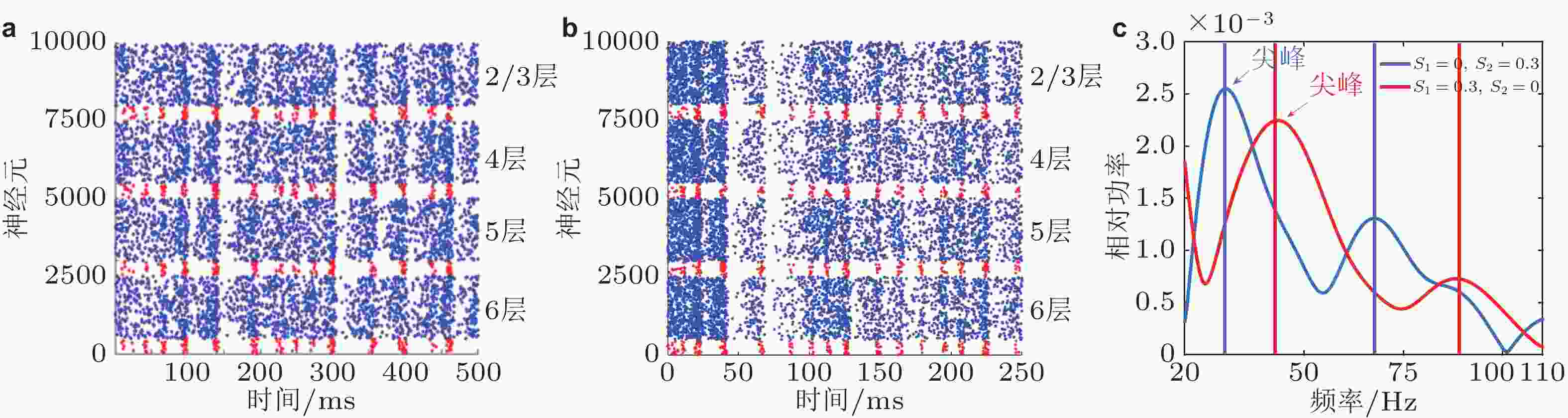
图 11 具有多层立柱结构的大规模复杂E/I神经网络产生的 Gamma 振荡. (a) 当外部输入为 S1 = 0.3、S2 = 0 时每层神经元的放电时刻斑图; (b) 当外部输入为 S1 = 0, S2 = 0.3时每层神经元的放电时刻斑图; 图 (a)(b)中红色点代表抑制性神经元的放电, 蓝色点代表兴奋性神经元的放电; (c) 分别对应于 (a) 和 (b) 的神经元群体活动的功率谱图 (Gu et al. 2021b)
图 12 具有多层立柱结构的大规模复杂E/I神经网络中Gamma 振荡的振荡频率对输入差异的依赖性. 红色和蓝色曲线分别代表中心刺激对比度增加 (增大S1、保持S2为0) 和周围刺激对比度增加 (增大S2、保持S1为0) 的调节情形 (Gu et al. 2021b)
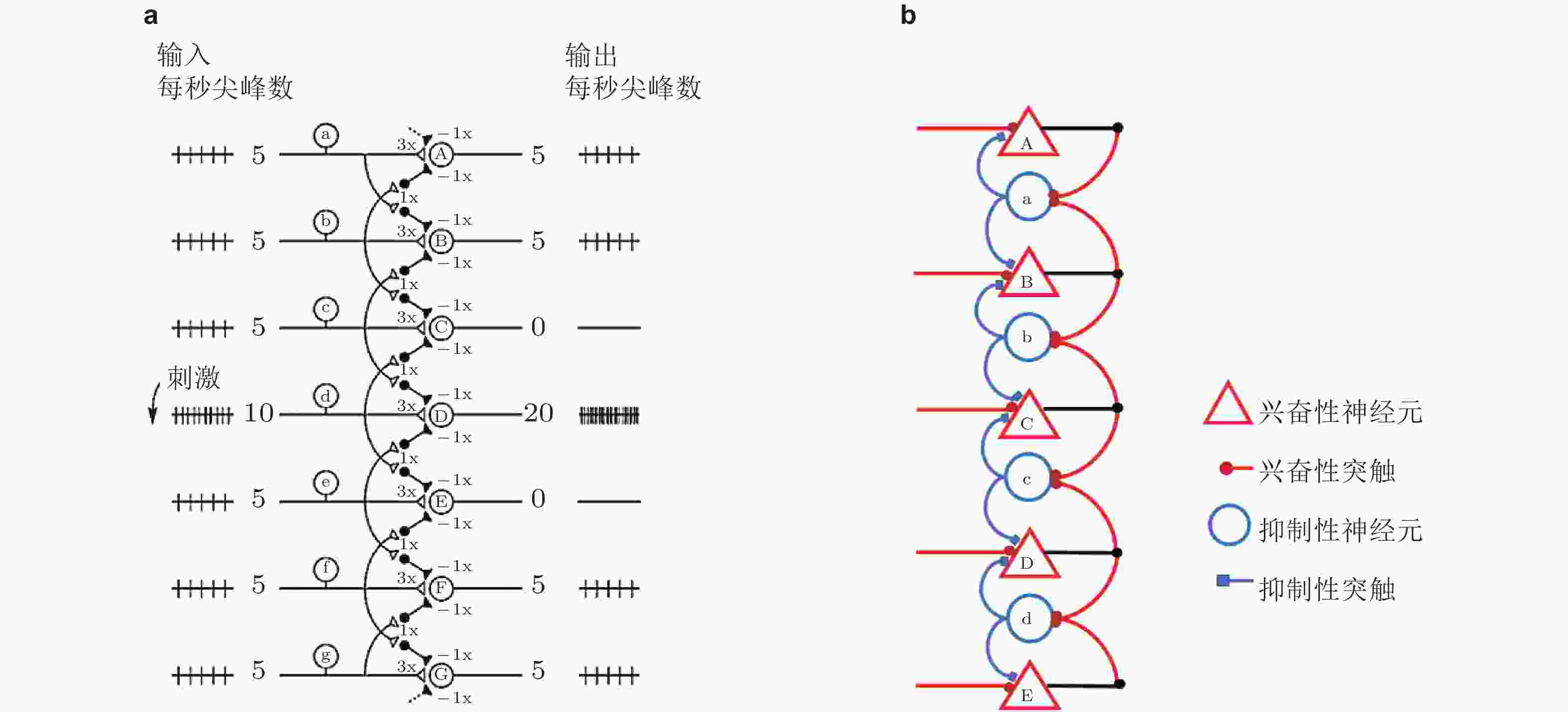
图 13 (a) 前馈神经网络结构 (Bear & Connors, 2004) . 标有字母A-G的圆圈表示兴奋性神经元, 小黑点表示抑制性神经元; (b) 侧抑制循环神经网络结构. 标有字母a-d的抑制性神经元收到来自侧向兴奋性神经元 (标有字母A-E) 的反馈, 并侧向抑制相邻的兴奋性神经元 (Han et al. 2018)
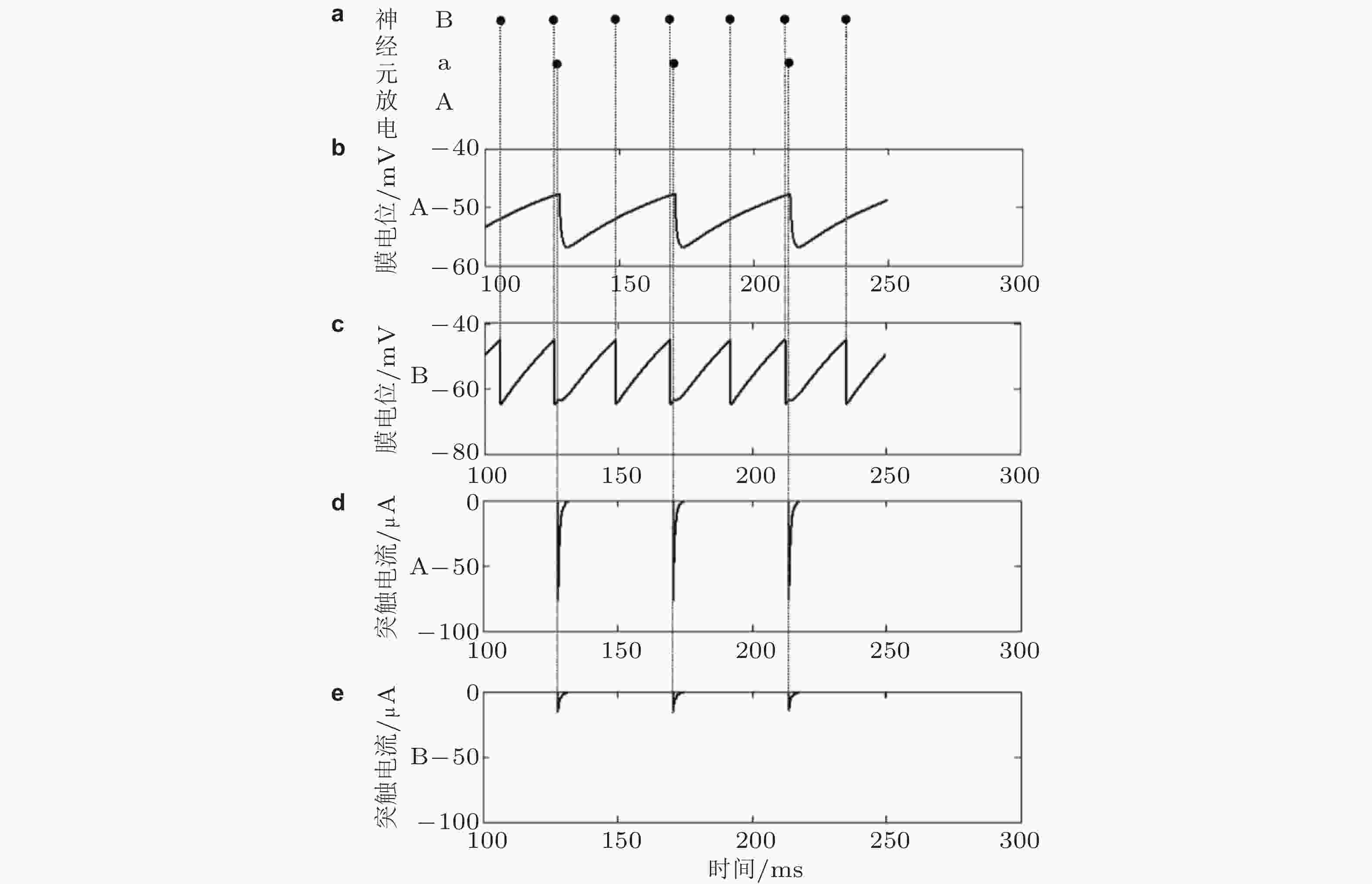
图 14 在兴奋性神经元“B”放电引起抑制性神经元“a”放电的情况下, 兴奋性神经元“A”和“B”的膜电位和接收的抑制性突触电流. (a) 神经元“B” “a”和“A”的放电, 黑点代表相应神经元的放电, 其中“A”未达到阈值电位而没有放电; (b) 神经元“A”的膜电位; (c) 神经元“B”的膜电位; (d) 神经元“A”接收的抑制性突触电流; (e) 神经元“B”接收的抑制性突触电流 (Han et al. 2018)
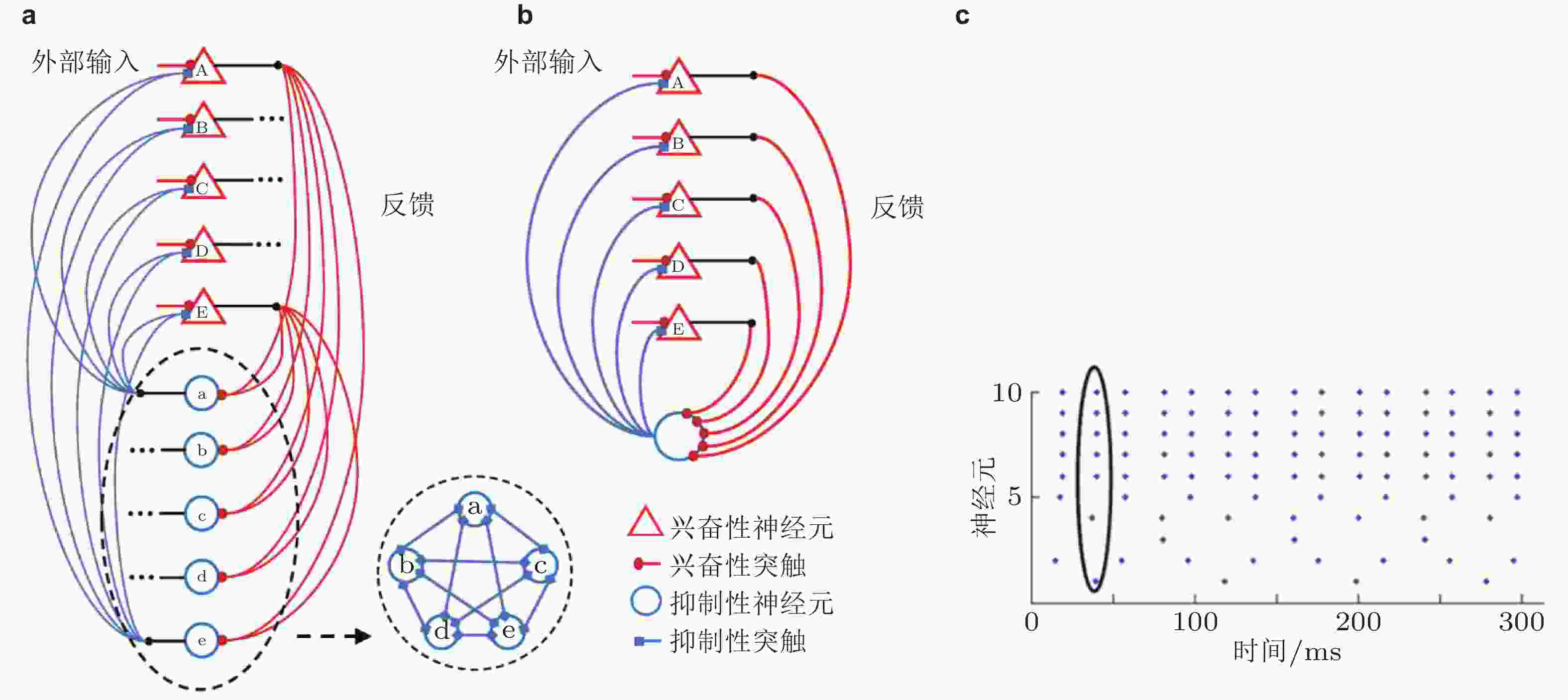
图 15 抑制性神经元同步放电的循环E/I神经网络及其放电模式. (a) 由5个兴奋性神经元和5个全连接的同步抑制性神经元 (在虚线椭圆中) 组成的简单E/I网络; (b) 与 (a) 等效的E/I网络; (c) 图 (a) 中E/I神经网络的同步放电模式, 索引为1 ~ 5的神经元表示兴奋性神经元 “A”-“E”, 索引为6 ~ 10的神经元表示抑制性神经元 (Han et al. 2018)
图 16 全局连接复杂E/I神经网络的结构 (Han et al. 2018)
图 17 (a) (c) E/I神经网络的同步和部分同步状态, N = 65, 其中神经元1 ~ 50是兴奋性神经元, 神经元51 ~ 65是抑制性神经元; (b) (d) E/I神经网络的放电率对比度, 图中方形线表示纯兴奋性神经元 (断开抑制性连接) 的归一化放电率的标准偏差, 星形线表示全局耦合的E/I神经网络中的兴奋性神经元的归一化放电率的标准偏差, 点线表示E/I神经网络和纯兴奋性神经元网络的放电率对比度增强倍数 (Han et al. 2018)
图 18 (a) 随机E/I网络结构. 该网络由一个大小为
$ {N}_{e} $ 的兴奋性神经元集群和一个大小为$ {N}_{i} $ 的抑制性神经元集群组成. 网络中任意神经元连接到其他神经元的概率为$ \rho $ . (b) 该网络产生的Gamma同步振荡 (Han et al. 2020)图 19 神经元输入无噪声情形下E/I神经网络的放电率对比度增强. (a) 纯兴奋性神经元网络里400个兴奋性神经元的放电率对比度可视化图像; (b) E/I神经网络中400个兴奋性神经元的放电率对比度可视化图像 (Han et al. 2020)
图 20 信息编码效能指标
$ {I}_{\mathrm{C}} $ 与以下参数的关系: 从抑制性神经元到兴奋性神经元的突触强度$ {g}^{\mathrm{I}\mathrm{E}} $ , 从兴奋性神经元到抑制性神经元的突触强度$ {g}^{\mathrm{E}\mathrm{I}} $ , 外部刺激的标准差$ {\delta }^{\mathrm{s}} $ (Han et al. 2020)表 1 侧循环抑制引起的局部放电率对比度增强 (Han et al. 2018)
不同的结构 放电率/归一化放电率 A B C D E 被分离的神经元 24/0.816 49/1.667 12/0.408 44/1.497 18/0.612 如图13(b) 连接 14/0.63 42/1.89 0/0 41/1.85 14/0.63 表 2 抑制性同步引起全局放电率对比度增强 (Han et al. 2018)
不同的结构 放电率/归一化放电率 A B C D E 被分离的神经元 24/0.816 49/1.667 12/0.408 44/1.497 18/0.612 如图15(a) 连接 14/0.686 44/2.157 0/0 36/1.765 8/0.392 -
Adjamian P, Hadjipapas A, Barnes G R, et al. 2008. Induced Gamma activity in primary visual cortex is related to luminance and not color contrast: an MEG study. Journal of Vision, 8: 1-7. doi: 10.1167/8.7.1 Adrian E D. 1935. Discharge frequencies in the cerebral and cerebellar cortex. Proceedings of the Physical Society, 83: 32-33. Adrian E D. 1942. Olfactory reactions in the brain of the hedgehog. The Journal of Physiology, 100: 459-473. doi: 10.1113/jphysiol.1942.sp003955 Aoyagi T, Kang Y, Terada N, et al. 2002. The role of Ca2+ dependent cationic current in generating Gamma frequency rhythmic bursts: modeling study. Neuroscience, 115: 1127-1138. doi: 10.1016/S0306-4522(02)00537-7 Bastos A M, Briggs F, Alitto H J, et al. 2014. Simultaneous recordings from the primary visual cortex and lateral geniculate nucleus reveal rhythmic interactions and a cortical source for Gamma-band. Journal of Neuroscience, 34: 7639-7644. doi: 10.1523/JNEUROSCI.4216-13.2014 Bartos M, Vida I, Frotscher M, et al. 2002. Fast synaptic inhibition promotes synchronized Gamma oscillations in hippocampal interneuron networks. Proceedings of the National Academy of Sciences of the United States of America, 99: 13222-13227. doi: 10.1073/pnas.192233099 Bartos M, Vida I, Jonas P. 2007. Synaptic mechanisms of synchronized Gamma oscillations in inhibitory interneuron networks. Nature Reviews Neuroscience, 8: 45-56. doi: 10.1038/nrn2044 Bathellier B, Lagier S, Faure P, et al. 2006. Circuit properties generating Gamma oscillations in a network model of the olfactory bulb. Journal of Neurophysiology, 95: 2678-2691. doi: 10.1152/jn.01141.2005 Bathellier B, Carleton A, Gerstner W. 2008. Gamma oscillations in a nonlinear regime: a minimal model approach using heterogeneous Integrate-and-Fire networks. Neural Computation, 20: 2973-3002. doi: 10.1162/neco.2008.11-07-636 Bauer M, Stenner M-P, Friston K J, Dolan R J. 2014. Attentional modulation of Alpha/Beta and Gamma oscillations reflect functionally distinct processes. Journal of Neuroscience, 34: 16117-16125. doi: 10.1523/JNEUROSCI.3474-13.2014 Bear M F, Connors B W, Paradiso M A. 2004. Neuroscience: Exploring the Brain. Beijing: High Education Press Bitzenhofer S H, Popplau J A, Hanganu-Opatz I. 2020. Gamma activity accelerates during prefrontal development. Elife, 9: e56795. doi: 10.7554/eLife.56795 Bland B, Brian H. 1986. The physiology and pharmacology of hippocampal formation Theta rhythms. Progrss in Neurobiology, 26: 1-54. doi: 10.1016/0301-0082(86)90019-5 Borgers C, Kopell N J. 2008. Gamma oscillations and stimulus selection. Neural Computation, 20: 383-414. doi: 10.1162/neco.2007.07-06-289 Borgers C, Epstein S, Kopell N J. 2008. Gamma oscillations mediate stimulus competition and attentional selection in a cortical network model. Proceedings of the National Academy of Sciences of the United States of America, 105: 18023-18028. doi: 10.1073/pnas.0809511105 Bouyer J, Montaron M, Rougeul A. 1981. Fast fronto-parietal rhythms during combined focused attentive behaviour and immobility in cat: Cortical and thalamic localizations. Electroencephalography and Clinical Neurophysiology, 51: 244-252. doi: 10.1016/0013-4694(81)90138-3 Bragin A, Jandó G, Nádasdy Z, et al. 1995. Gamma (40-100 Hz) oscillation in the hippocampus of the behaving rat. Journal of Neuroscience, 15: 47-60. doi: 10.1523/JNEUROSCI.15-01-00047.1995 Bremer F. 1958. Cerebral and cerebellar potential. Physiological Reviews, 58: 357-388. Brette R, Rudolph M, Carnevale T, et al. 2007. Simulation of networks of spiking neurons: a review of tools and strategies. Journal of Computational Neuroscience, 23: 349-398. doi: 10.1007/s10827-007-0038-6 Brunet N M, Bosman C A, Vinck M, et al. 2014. Stimulus repetition modulates Gamma-band synchronization in primate visual cortex. Proceedings of the National Academy of Sciences, 111: 3626-3631. doi: 10.1073/pnas.1309714111 Buzsáki G. 2006. Rhythms of the Brain. Oxford University Press Buzsáki G, Draguhn A. 2004. Neuronal oscillations in cortical networks. Science, 304: 1926-1929. doi: 10.1126/science.1099745 Buzsáki G, Horváth Z, Urioste R, et al. 1992. High-frequency network oscillation in the hippocampus. Science, 256: 1025-1027. doi: 10.1126/science.1589772 Buzśaki G, Wang X J. 2012. Mechanisms of Gamma oscillations. Annual Review of Neuroscience, 35: 203-225. doi: 10.1146/annurev-neuro-062111-150444 Canolty R T, Edwards E, Dalal S S, et al. 2006. High Gamma power is phase-locked to Theta oscillations in human neocortex. Science, 313: 1626-1628. doi: 10.1126/science.1128115 César, Rennó-Costa, Garcia D, et al. 2019. Regulation of Gamma-frequency oscillation by feedforward inhibition: A computational modeling study. Hippocampus, 29: 957-970. doi: 10.1002/hipo.23093 Chapeau-Blondeau F, Chambet N. 1995. Synapse models for neural networks: From ion channel kinetics to multiplicative coefficient wij. Neural Computation, 7: 713-734. doi: 10.1162/neco.1995.7.4.713 Cole S R, Voytek B. 2017. Brain oscillations and the importance of waveform shape. Trends in Cognitive Sciences, 21: 137-149. doi: 10.1016/j.tics.2016.12.008 Dayan P, Abbott L F. 2001. Theoretical Neuroscience: Computational and Mathematical Modeling of Neural Systems. Mit Press Demiralp T, Bayraktaroglu Z, Lenz D, et al. 2007. Gamma amplitudes are coupled to Theta phase in human EEG during visual perception. International Journal of Psychophysiology, 64: 24-30. doi: 10.1016/j.ijpsycho.2006.07.005 Destexhe A, Mainen Z F, Sejnowski T J. 1998. Kinetic models of synaptic transmission. Methods in Neuronal Modeling, 2: 1-25. Doesburg S M, Roggeveen A B, Kitajo K, et al. 2008. Large-scale Gamma-band phase synchronization and selective attention. Cerebral Cortex, 18: 386-396. doi: 10.1093/cercor/bhm073 Fan J, Byrne J, Worden M S, et al. 2007. The relation of brain oscillations to attentional networks. Journal of Neuroscience, 27: 6197-6206. doi: 10.1523/JNEUROSCI.1833-07.2007 Fitzgerald P J, Watson B O. 2018. Gamma oscillations as a biomarker for major depression: an emerging topic. Translational Psychiatry, 8: 177. doi: 10.1038/s41398-018-0239-y FitzHugh R. 1961. Impulses and physiological states in theoretical models of nerve membrane. Biophysical Journal, 1: 445-466. doi: 10.1016/S0006-3495(61)86902-6 Freeman W J. 1975. Mass Action in the Nervous System. Academic Press Frien A, Eckhorn R, Bauer R, et al. 1994. Stimulus-specific fast oscillations at zero phase between visual areas V1 and V2 of awake monkey. NeuroReport, 5: 2273-2277. doi: 10.1097/00001756-199411000-00017 Fries P. 2005. A mechanism for cognitive dynamics: Neuronal communication through neuronal coherence. Trends in Cognitive Sciences, 9: 474-480. doi: 10.1016/j.tics.2005.08.011 Fries P. 2015. Rhythms for cognition: Communication through coherence. Neuron, 88: 220-235. doi: 10.1016/j.neuron.2015.09.034 Fries P, Nikolić D, Singer W. 2007. The Gamma cycle. Trends in Neurosciences, 30: 309-316. doi: 10.1016/j.tins.2007.05.005 Fries P, Reynolds J H, Rorie A E, et al. 2001. Modulation of oscillatory neuronal synchronization by selective visual attention. Science, 291: 1560-1563. doi: 10.1126/science.1055465 Gazit T, Friedman A, Lax E, et al. 2015. Programmed deep brain stimulation synchronizes VTA gamma band field potential and alleviates depressive-like behavior in rats. Neuropharmacology, 91: 135-141. doi: 10.1016/j.neuropharm.2014.12.003 Glass L. 2001. Synchronization and rhythmic processes in physiology. Nature, 410: 277-284. doi: 10.1038/35065745 Gieselmann M A, Thiele A. 2008. Comparison of spatial integration and surround suppression characteristics in spiking activity and the local field potential in macaque V1. European Journal of Neuroscience, 28: 447-459. doi: 10.1111/j.1460-9568.2008.06358.x Gray C M, Knig P, Engel A K, et al. 1989. Oscillatory responses in cat visual cortex exhibit inter-columnar synchronization which reflects global stimulus properties. Nature, 338: 334-337. doi: 10.1038/338334a0 Gregory S, Fusca M, Rees G, et al. 2016. Gamma frequency and the spatial tuning of primary visual cortex. PLoS One, 11: e0157374. doi: 10.1371/journal.pone.0157374 Grossberg S, Versace M. 2008. Spikes, synchrony, and attentive learning by laminar thalamocortical circuits. Brain Research, 1218: 278-312. doi: 10.1016/j.brainres.2008.04.024 Gruber T, Müller M M, Keil A, et al. 1999. Selective visual-spatial attention alters induced Gamma band responses in the human EEG. Clinical Neurophysiology, 110: 2074-2085. doi: 10.1016/S1388-2457(99)00176-5 Gu X C, Han F, Wang Z J, et al. 2019. Dependency of Gamma oscillations in E/I neuronal network on illumination contrast of external stimulus. Theoretical & Applied Mechanics Letters, 9: 21-27. Gu X C, Han F, Wang Z J. 2021a. Dependency analysis of frequency and strength of Gamma oscillations on input difference between excitatory and inhibitory neurons. Cognitive Neurodynamics, 15: 501-515. doi: 10.1007/s11571-020-09622-5 Gu X C, Han F, Wang Z J, Kashif K, et al. 2021b. Enhancement of Gamma oscillations in E/I neural networks by increase of difference between external inputs. Electronic Research Archive, 29: 3227-3241. doi: 10.3934/era.2021035 Han F, Gu X, Wang Z J, et al. 2018. Global firing rate contrast enhancement in E/I neuronal networks by recurrent synchronized inhibition. Chaos, 28: 106324. doi: 10.1063/1.5037207 Han F, Wang Z J, Fan H, et al. 2020. High-frequency synchronization improves firing rate contrast and information transmission efficiency in E/I neuronal networks. Neural Plasticity, 2020: 8823111. Han F, Wang Z J, Fan H. 2017. Determine neuronal tuning curves by exploring optimum firing rate distribution for information efficiency. Frontiers in Computational Neuroscience, 11: 10. Hauck M, Lorenz J, Engel A K. 2007. Attention to painful stimulation enhances γ-band activity and synchronization in human sensorimotor cortex. Journal of Neuroscience, 27: 9270-9277. doi: 10.1523/JNEUROSCI.2283-07.2007 Henrie J A, Kang K, Shapley R. 2005. Stimulus size affects the LFP spectral contents in primate V1. Society for Neuroscience, 35: 18. Henrie J A, Shapley R. 2005. LFP power spectra in v1 cortex: The graded effect of stimulus contrast. Journal of Neurophysiology, 94: 479-490. doi: 10.1152/jn.00919.2004 Herrmann C S, Murray M M, Ionta S, et al. 2016. Shaping intrinsic neural oscillations with periodic stimulation. Journal of Neuroscience, 36: 5328-5337. doi: 10.1523/JNEUROSCI.0236-16.2016 Hindmarsh J L, Rose R M. 1982. A model of the nerve impulse using two first-order differential equations. Nature, 296: 162-164. doi: 10.1038/296162a0 Hipp J F, Engel A K, Siegel M. 2011. Oscillatory synchronization in large-scale cortical networks predicts perception. Neuron, 69: 387-396. doi: 10.1016/j.neuron.2010.12.027 Hodgkin A L, Huxley A F. 1952. Currents carried by sodium and potassium ions through the membrane of the giant axon of Loligo. Journal of Physiology, 116: 449. doi: 10.1113/jphysiol.1952.sp004717 Howard M W, Rizzuto D S, Caplan J B, et al. 2003. Gamma oscillations correlate with working memory load in humans. Cerebral Cortex, 13: 1369-1374. doi: 10.1093/cercor/bhg084 Hutt A, Mierau A, Lefebvre J. 2016. Dynamic control of synchronous activity in networks of spiking neurons. PLoS ONE, 11: e0161488. doi: 10.1371/journal.pone.0161488 Huxter J, Burgess N, O’Keefe1 J. 2003. Independent rate and temporal coding in hippocampal pyramidal cells. Nature, 425: 828-832. doi: 10.1038/nature02058 Izhikevich E M. 2003. Simple model of spiking neurons. IEEE Transactions on Neural Networks and Learning Systems, 14: 1569-1572. doi: 10.1109/TNN.2003.820440 Jadi M P, Behrens M M, Sejnowski T J. 2016. Abnormal Gamma oscillations in N-methyl-D-aspartate receptor hypofunction models of schizophrenia. Biological Psychiatry, 79: 716-726. doi: 10.1016/j.biopsych.2015.07.005 Jadi M P, Sejnowski T J. 2014a. Cortical oscillations arise from contextual interactions that regulate sparse coding. Proceedings of the National Academy of Sciences of the United States of America, 111: 6780-6785. doi: 10.1073/pnas.1405300111 Jadi M P, Sejnowski T J. 2014b. Regulating cortical oscillations in an inhibition-stabilized network. Proceedings of the IEEE, 102: 830-842. doi: 10.1109/JPROC.2014.2313113 Jasper H, Penfield W. 1949. Electrocorticograms in man: Effect of voluntary movement upon the electrical activity of the precentral gyrus. European Archives of Psychiatry and Clinical Neuroscience, 183: 163-174. Jensen O, Tesche C D. 2010. Frontal Theta activity in humans increases with memory load in a working memory task. European Journal of Neuroscience, 15: 1395-1399. Jerbi K, Hamamé C M, Ossandón T, et al. 2008. Role of posterior parietal Gamma activity in planning prosaccades and antisaccades. Journal of Neuroscience, 28: 13713-13715. doi: 10.1523/JNEUROSCI.4896-08.2008 Jia X, Tanabe S, KohnA. 2013a. Gamma and the coordination of spiking activity in early visual cortex. Neuron, 77: 762-774. doi: 10.1016/j.neuron.2012.12.036 Jia X, Xing D, Kohn A. 2013b. No consistent relationship between Gamma power and peak frequency in macaque primary visual cortex. Journal of Neuroscience, 33: 17-25. doi: 10.1523/JNEUROSCI.1687-12.2013 Kahana M J, Sekuler R, Caplan J B, et al. 1999. Human Theta oscillations exhibit task dependence during virtual maze navigation. Nature, 399: 781-784. doi: 10.1038/21645 Kaiser J, Bühler M, Lutzenberger W. 2004. Magnetoencephalographic Gamma-band responses to illusory triangles in humans. NeuroImage, 23: 551-560. doi: 10.1016/j.neuroimage.2004.06.033 Kang K, Shelley M, Henrie J A, et al. 2010. LFP Spectral peaks in V1 cortex: network resonance and cortico-cortical feedback. Journal of Computational Neuroscience, 29: 495-507. doi: 10.1007/s10827-009-0190-2 Khalid A, Kim BS, Seo BA, et al. 2016. Gamma oscillation in functional brain networks is involved in the spontaneous remission of depressive behavior induced by chronic restraint stress in mice. BMC Neurosci, 17: 4. Kim J, Bertalmio M. 2015. Investigating the effect of lateral inhibition in the retinal circuitry on lightness contrast and assimilation: a model study. Journal of Vision, 15: 633. doi: 10.1167/15.12.633 Koch S P, Werner P, Steinbrink J, et al. 2009. Stimulus-induced and state-dependent sustained Gamma activity is tightly coupled to the hemodynamic response in humans. Journal of Neuroscience, 29: 13962-13970. doi: 10.1523/JNEUROSCI.1402-09.2009 Kopell N J, Gritton H J, Whittington M A, Kramer M A. 2014. Beyond the connectome: the dynome. Neuron, 83: 1319-1328. doi: 10.1016/j.neuron.2014.08.016 Kostal L, Lansky P. 2013. Information capacity and its approximations under metabolic cost in a simple homogeneous population of neurons. Biosystems, 112: 265-275. doi: 10.1016/j.biosystems.2013.03.019 Kropotov J D. 2009. Quantitative EEG, Event-Related Potentials and Neurotherapy. Academic Press. Lapicque L. 1907. Recherches quantitatives sur l'excitation electrique des nerfs traitée comme une polarization. Journal of Physiology and Pathology General, 9: 620-635. Lefebvre J, Hutt A, Knebel J F, et al. 2015. Stimulus statistics shape oscillations in nonlinear recurrent neural networks. Journal of Neuroscience, 35: 2895-2903. doi: 10.1523/JNEUROSCI.3609-14.2015 Li K T, Liang J, Zhou C. 2021. Gamma oscillations facilitate effective learning in excitatory-inhibitory balanced neural circuits. Neural Plasticity, 7: 1-18. Lu Y, Sarter M, Zochowski M, et al. 2020. Phasic cholinergic signaling promotes emergence of local Gamma rhythms in excitatory–inhibitory networks. European Journal of Neuroscience, 52: 3545-3560. doi: 10.1111/ejn.14744 Magazzini L, Singh K D. 2018. Spatial attention modulates visual Gamma oscillations across the human ventral stream. NeuroImage, 166: 219-229. doi: 10.1016/j.neuroimage.2017.10.069 Masuda N. 2009. Selective population rate coding: A possible computational role of Gamma oscillations in selective attention. Neural Computation, 21: 3335-3362. doi: 10.1162/neco.2009.09-08-857 Mcclelland J L, Rumelhard D E. 1986. Exploration in Parallel Distributed Processing, A Handbook of Models, Programs, and Exercises. Cambridge: MIT Press McCulloch W S, Pitts W. 1943. A logical calculus of the ideas immanent in neurons activity. Bulletin of Mathematical Biophysics, 5: 115-133. doi: 10.1007/BF02478259 Morrison A, Aertsen A, Diesmann M. 2007. Spike-timing dependent plasticity in balanced random networks thanks. Neural Computation, 19: 1437-1467. doi: 10.1162/neco.2007.19.6.1437 Moujahid A, d'Anjou A, Torrealdea F J, Torrealdea F. 2011. Energy and information in Hodgkin-Huxley neurons. Physical Review E, 83: 031912. doi: 10.1103/PhysRevE.83.031912 Mountcastle V B. 1997. The columnar organization of the neocortex. Brain, 120: 701-722. doi: 10.1093/brain/120.4.701 Muthukumaraswamy S D, Singh K D. 2013. Visual Gamma oscillations: the effects of stimulus type, visual field coverage and stimulus motion on meg and EEG recordings. NeuroImage, 69: 223-230. doi: 10.1016/j.neuroimage.2012.12.038 Neymotin S A, Lee H, Park E, et al. 2011. Emergence of physiological oscillation frequencies in a computer model of neocortex. Frontiers in Computational Neuroscience, 5: 19. Oliveira L D R, Gomes R M, Santos B A, et al. 2019. Effects of the parameters on the oscillation frequency of Izhikevich spiking neural networks. Neurocomputing, 337: 251-261. doi: 10.1016/j.neucom.2019.01.071 Onslow A C E, Jones M W, Bogacz R. 2014. A canonical circuit for generating phase-amplitude coupling. PLoS ONE, 9: e102591. doi: 10.1371/journal.pone.0102591 Orekhova E V, Butorina A V, Sysoeva O V, et al. 2015. Frequency of Gamma oscillations in humans is modulated by velocity of visual motion. Journal of Neurophysiology, 114: 244-255. doi: 10.1152/jn.00232.2015 Osipova D, Takashima A, Oostenveld R, et al. 2006. Theta and Gamma oscillations predict encoding and retrieval of declarative memory. Journal of Neuroscience, 26: 7523-7531. doi: 10.1523/JNEUROSCI.1948-06.2006 Panzeri S, Brunel N, Logothetis N K, et al. 2010. Sensory neural codes using multiplexed temporal scales. Trends in Neurosciences, 33: 111-120. doi: 10.1016/j.tins.2009.12.001 Peng X, Wang Z, Han F, et al. 2018. A novel time-event-driven algorithm for simulating spiking neural networks based on circular array. Neurocomputing, 292: 121-129. doi: 10.1016/j.neucom.2018.02.085 Perry G, Randle J M, Koelewijn L, et al. 2015. Linear tuning of Gamma amplitude and frequency to luminance contrast: evidence from a continuous mapping paradigm. PLoS One, 10: e0124798. doi: 10.1371/journal.pone.0124798 Peter A, Stauch B J, Shapcott K, et al. 2021. Stimulus-specific plasticity of macaque V1 spike rates and Gamma. Cell Reports, 37: 110086. doi: 10.1016/j.celrep.2021.110086 Petersen C C H, Sakmann B. 2013. Functionally independent columns of rat somatosensory barrel cortex revealed with voltage-sensitive dye imaging. Journal of Neuroscience, 21: 8435-8446. Pesaran B, Pezaris J, Sahani M, et al. 2002. Temporal structure in neuronal activity during working memory in macaque parietal cortex. Nature Neuroscience, 5: 805-811. doi: 10.1038/nn890 Rall W. 1967. Distinguishing theoretical synaptic potentials computed for different soma-dendritic distributions of synaptic input. Journal of Neurophysiology, 30: 1138-1168. doi: 10.1152/jn.1967.30.5.1138 Ray S, Maunsell J H R. 2010. Differences in Gamma frequencies across visual cortex restrict their possible use in computation. Neuron, 67: 885-896. doi: 10.1016/j.neuron.2010.08.004 Ray S, Maunsell J H R. 2011. Different origins of Gamma rhythm and high-Gamma activity in macaque visual cortex. PLoS Biology, 9: e1000610. doi: 10.1371/journal.pbio.1000610 Richard A Y. 1987. The gaussian derivative model for spatial vision: i. retinal mechanisms. Spatial Vision, 2: 273-293. doi: 10.1163/156856887X00222 Rodieck R W. 1965. Quantitative analysis of cat retinal ganglion cell response to visual stimuli. Vision Research, 5: 583-601. doi: 10.1016/0042-6989(65)90033-7 Rodieck R W. 1975. Analysis of receptive fields of cat retinal ganglion cells. Journal of Neurophysiology, 28: 833-849. Sacerdote L, Giraudo M T. 2013. Stochastic integrate and fire models: a review on mathematical methods and their applications. Quantitative Biology, 2058: 99-148. Saleem A B, Lien A D, Krumin M, et al. 2017. Subcortical source and modulation of the narrowband Gamma oscillation in mouse visual cortex. Neuron, 93: 315-322. doi: 10.1016/j.neuron.2016.12.028 Sato Y, Ochi A, Mizutani T, Otsubo H. 2019. Low entropy of interictal Gamma oscillations is a biomarker of the seizure onset zone in focal cortical dysplasia type II. Epilepsy & Behavior, 96: 155-159. Schnitzler A, Gross J. 2005. Normal and pathological oscillatory communication in the brain. Nature Reviews Neuroscience, 6: 285-296. doi: 10.1038/nrn1650 Schroeder C E, Lakatos P. 2009. Low-frequency neuronal oscillations as instruments of sensory selection. Trends in Neurosciences, 32: 9-18. doi: 10.1016/j.tins.2008.09.012 Schwarzkopf D S, Robertson D J, Song C, et al. 2012. The Frequency of visually induced Gamma-band oscillations depends on the size of early human visual cortex. Journal of Neuroscience, 32: 1507-1512. doi: 10.1523/JNEUROSCI.4771-11.2012 Sengupta B, Laughlin S B, Niven J E. 2014. Consequences of converting graded to action potentials upon neural information coding and energy efficiency. PLoS Computational Biology, 10: e1003439. doi: 10.1371/journal.pcbi.1003439 Senkowski D, Schneider T R, Foxe J J, Engel A K. 2008. Crossmodal binding through neural coherence: implications for multisensory processing. Trends in Neurosciences, 31: 401-409. doi: 10.1016/j.tins.2008.05.002 Sherfey J, Ardid S, Miller E K, et al. 2020. Prefrontal oscillations modulate the propagation of neuronal activity required for working memory. Neurobiology of Learning and Memory, 173: 107228. doi: 10.1016/j.nlm.2020.107228 Siegel M, Donner T H, Engel A K. 2012. Spectral fingerprints of large-scale neuronal interactions. Nature Reviews Neuroscience, 13: 121-134. doi: 10.1038/nrn3137 Siegel M, Donner T H, Oostenveld R, et al. 2008. Neuronal synchronization along the dorsal visual pathway reflects the focus of spatial attention. Neuron, 60: 709-719. doi: 10.1016/j.neuron.2008.09.010 Singer W, Gray C M. 1995. Visual feature integration and the temporal correlation hypothesis. Annual Review of Neuroscience, 18: 555-586. doi: 10.1146/annurev.ne.18.030195.003011 Sokolov A, Lutzenberger W, Pavlova M, et al. 1999. Gamma-band meg activity to coherent motion depends on task-driven attention. NeuroReport, 10: 1997-2000. doi: 10.1097/00001756-199907130-00001 Storchi R, Bedford R A, Martial F P, et al. 2017. Modulation of fast narrowband oscillations in the mouse retina and dLGN according to background light intensity. Neuron, 93: 299-307. doi: 10.1016/j.neuron.2016.12.027 Swettenham J B, Muthukumaraswamy S D, Singh K D. 2009. Spectral properties of induced and evoked Gamma oscillations in human early visual cortex to moving and stationary stimuli. Journal of Neurophysiology, 102: 1241-1253. doi: 10.1152/jn.91044.2008 Tan L L, Oswald M J, Kuner B, 2021. Neurobiology of brain oscillations in acute and chronic pain. Trends in Neurosciences, 44: 629-642. Tiesinga P, Sejnowski T J. 2009. Cortical enlightenment: are attentional Gamma oscillations driven by ING or PING. Neuron, 63: 727-732. doi: 10.1016/j.neuron.2009.09.009 Trujillo C A, Gao R, Negraes P D. et al. 2019. Complex oscillatory waves emerging from cortical organoids model early human brain network development. Cell Stem Cell, 25: 1-12. Valley M T, Firestein S A, 2008. Lateral look at olfactory bulb lateral inhibition. Neuron, 59: 682-684. Vanderwolf C H. 1969. Hippocampal electrical activity and voluntary movement in the rat. Electroencephalography and Clinical Neurophysiology, 26: 407-418. doi: 10.1016/0013-4694(69)90092-3 Vida I, Bartos M, Jonas P. 2006. Shunting inhibition improves robustness of Gamma oscillations in hippocampal interneuron networks by homogenizing firing rates. Neuron, 49: 107-117. doi: 10.1016/j.neuron.2005.11.036 Vinck M, Batista-Brito R, Knoblich U, et al. 2015. Arousal and locomotion make distinct contributions to cortical activity patterns and visual encoding. Neuron, 86: 740-754. doi: 10.1016/j.neuron.2015.03.028 Wallace E, Benayoun M, Van Drongelen W, et al. 2011. Emergent oscillations in networks of stochastic spiking neurons. PLoS ONE, 6: e14804. doi: 10.1371/journal.pone.0014804 Wang X J. 2010. Neurophysiological and computational principles of cortical rhythms in cognition. Physiological Reviews, 90: 1195-1268. doi: 10.1152/physrev.00035.2008 Wang X J, Buzsáki G. 1996. Gamma oscillation by synaptic inhibition in a hippocampal interneuronal network model. Journal of Neuroscience, 16: 6402-6413. doi: 10.1523/JNEUROSCI.16-20-06402.1996 Wang Y H, Wang R B, Xu X Y. 2017. Neural energy supply-consumption properties based on Hodgkin-Huxley model. Neural Plasticity, 2017: 6207141. Wang Z J, Han F, Aihara K. 2011. Three synaptic components contributing to robust network synchronization. Physical Review E, 83: 051905. doi: 10.1103/PhysRevE.83.051905 Wang Z J, Peng X, Han F, et al. 2020. A novel parallel clock-driven algorithm for simulation of neuronal networks based on virtual synapse. Simulation:Transactions of the Society for Modeling and Simulation International, 96: 415-427. doi: 10.1177/0037549720903804 Whittington M A, Traub R D, Jefferys J G R. 1995. Synchronized oscillations in interneuron networks driven by metabotropic glutamate receptor activation. Nature, 373: 612-615. doi: 10.1038/373612a0 Wildie M, Shanahan M. 2012. Establishing communication between neuronal populations through competitive entrainment. Frontiers in Computational Neuroscience, 5: 62. Xing D, Yeh C, Burns S, et al. 2012. Laminar analysis of visually evoked activity in the primary visual cortex. Proceedings of the National Academy of Sciences of the United States of America, 109: 13871-13876. Zhu Z, Wang R, Zhu F. 2018. The energy coding of a structural neural network based on the Hodgkin-Huxley model. Frontiers in Neuroscience, 12: 122. doi: 10.3389/fnins.2018.00122 -



 下载:
下载: